TGA makes final decision on paracetamol pack sizes
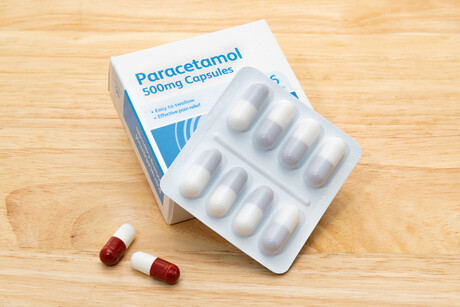
The Therapeutic Goods Administration (TGA) has published a final decision to reduce the maximum size of packs for various paracetamol products.
Each year in Australia around 225 people are hospitalised with liver injury and 50 Australians die from paracetamol overdose, with rates of intentional overdose highest among adolescents and young adults, the TGA said. This decision aims to reduce the harm from intentional overdose.
From 1 February 2025, new restrictions on paracetamol will:
- reduce the maximum size of packs available for general sale (eg, supermarkets and convenience stores) from 20 to 16 tablets or capsules
- reduce the maximum size of packs available in pharmacies without the supervision of a pharmacist (ie, ‘Pharmacy Only’ packs) from 100 to 50 tablets or capsules
- make other pack sizes of up to 100 tablets or capsules available only under the supervision of a pharmacist (‘Pharmacist Only’ medicines).
Paracetamol tablets and capsules for both general and Pharmacy Only sale will also be required to be in blister packaging. The maximum size of Pharmacy Only packs of individually wrapped powders or sachets of granules containing paracetamol will also be reduced in line with tablet and capsule packs.
Access to liquid, modified-release and Schedule 4 (Prescription Only) paracetamol is not affected by this decision.
The final decision to change the scheduling of paracetamol in the Poisons Standard has been made following consultation in February–March 2023 on an interim decision that proposed changes to maximum paracetamol pack sizes and packaging. The final decision carefully considered the feedback and balances the need for some restrictions to pack sizes while ensuring continued access to larger packs for those people who need regular pain relief.
The decision takes into account:
- an independent expert report commissioned by the TGA that examined the incidence of serious injury and death from intentional paracetamol overdose
- advice received from the Advisory Committee on Medicines Scheduling (ACMS)
- submissions from two rounds of consultation from individuals and organisations representing consumers, healthcare practitioners and industry.
To further minimise the harm from paracetamol overdose, the TGA is encouraging retailers such as supermarkets to restrict sales to a single pack at a time. The TGA is also encouraging consumers not to stockpile paracetamol in their home and to appropriately store paracetamol and other medicines. To allow manufacturers and retail outlets sufficient time to adjust, the decision takes effect from 1 February 2025.
Several industry experts have reacted to the news.
“Intentional overdose in adolescents and young adults is frequently driven by what is available in the home, and therefore reducing maximum size of packs will likely reduce risk of intentional paracetamol doses,” said Dr Renly Lim, pharmacist and Senior Research Fellow in the Quality Use of Medicine and Pharmacy Research Centre, University of South Australia.
“Medical treatment is needed within two to six hours after the paracetamol overdose. If treatment is delayed after ingesting high doses of paracetamol, there is a risk of serious liver injury, and sometimes death. Thus it is best to keep smaller quantities in the house, particularly where households may have members at risk of self-harm,” Lim said.
Professor Nicholas Buckley, clinical pharmacologist from the School of Medical Sciences, The University of Sydney, said the TGA was making a trade-off.
“Reducing pack sizes and making medicines prescription only have both repeatedly been proven effective methods to reduce severe poisonings. However, all scheduling decisions are a trade-off — in this case the TGA is making a trade-off between providing easy access to an affordable pain killer for the whole population and trying to reduce the severity of the 7000 paracetamol poisonings each year.
“Currently around 200 people suffer severe liver injury and around 20 die each year, and poisons centre data continue to show rapidly rising rates of deliberate self-poisoning in young girls and women.”
Some experts praised the TGA for balancing community needs with the potential for paracetamol overdose.
“While limiting deaths from paracetamol poisoning is important, these medicines are commonly used to manage pain in the community, and there is a need to ensure people living with pain are not adversely affected by new restrictions,” said Ria Hopkins, Senior Research Officer and PhD Candidate for the National Drug and Alcohol Research Centre, UNSW Sydney.
“The TGA’s decision appears to have taken into consideration the need to balance limiting harms from paracetamol while still ensuring these medicines remain accessible to Australians to manage pain,” she said.
Charges laid following patient death at The Royal Melbourne Hospital
After a patient admitted to The Royal Melbourne Hospital for an emergency mental health...
PASA Health & Aged Care Procurement Conference returns in June
Running 3–4 June 2026, the 11th Annual PASA Health & Aged Care Procurement Conference...
Stabbing of on-duty paramedic leads to attempted murder charge
Victoria Police charged a 32-year-old man in relation to the stabbing of an on-duty paramedic...




![[New Zealand] Transform from Security Awareness to a Security Culture: A Vital Shift for SMB Healthcare — Webinar](https://d1v1e13ebw3o15.cloudfront.net/data/89856/wfmedia_thumb/..jpg)
![[Australia] Transform from Security Awareness to a Security Culture: A Vital Shift for SMB Healthcare — Webinar](https://d1v1e13ebw3o15.cloudfront.net/data/89855/wfmedia_thumb/..jpg)



